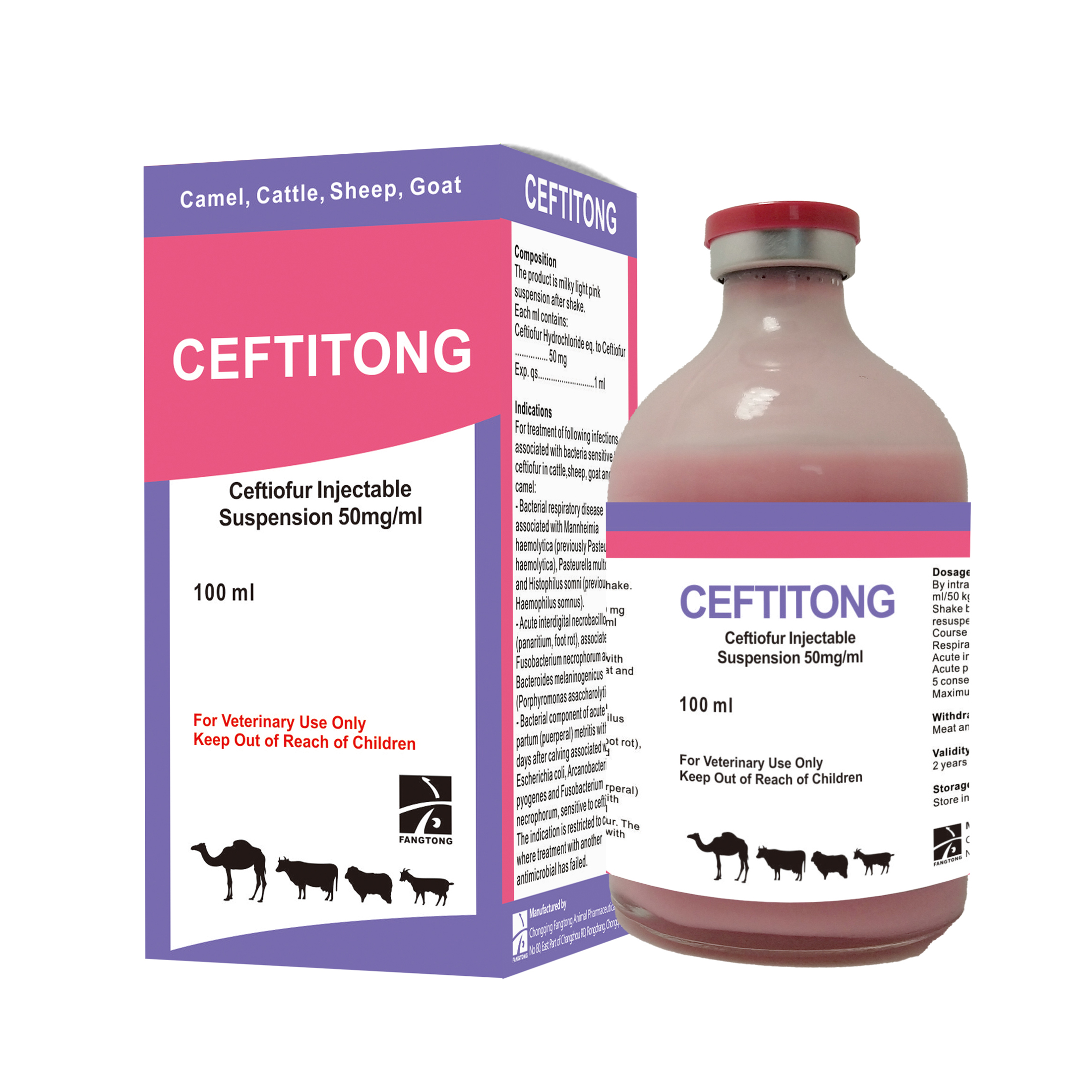
Composition
The product is milky light pink suspension after shake. Each ml contains:
Ceftiofur Hydrochloride eq. to Ceftiofur …………50 mg
Exp. qs……………………………………………1 ml
Indications
For treatment of following infections associated with bacteria sensitive to ceftiofur in cattle, sheep, goat and camel:
- Bacterial respiratory disease associated with Mannheimia haemolytica (previously Pasteurella haemolytica), Pasteurella multocida and Histophilus somni (previously Haemophilus somnus).
- Acute interdigital necrobacillosis (panaritium, foot rot), associated with Fusobacterium necrophorum and Bacteroides melaninogenicus (Porphyromonas asaccharolytica).
- Bacterial component of acute post-partum (puerperal) metritis within 10 days after calving associated with Escherichia coli, Arcanobacterium pyogenes and Fusobacterium necrophorum, sensitive to ceftiofur. The indication is restricted to cases where treatment with another antimicrobial has failed.
Contraindications
Do not use in cases of hypersensitivity to ceftiofur and other β-lactam antibiotics.
Do not inject intravenously.
Do not use in cases where resistance to other cephalosporins or beta-lactam antibiotics has occurred.
Do not use in poultry (including eggs) due to risk of spread of antimicrobial resistance to humans.
Adverse reactions
Hypersensitivity reactions unrelated to dose can occur. Allergic reactions (e.g. skin reactions, anaphylaxia) may occasionally occur. In case of the occurrence of allergic reaction the treatment should be withdrawn.
In cattle, mild inflammatory reactions at the injection site, such as tissue oedema and discoloration of the subcutaneous tissue and/or fascial surface of the muscle may be observed. Clinical resolution is reached in most animals by 10 days after injection although slight tissue discoloration may persist for 28 days or more.
Dosage and administration
By subcutaneous or intramuscular injection at dosage rate of 1 mg/kg (i.e. 1 ml/50 kg) b.w./day.
Shake before use until the product is adequately resuspended.
Course of treatment –
Respiratory disease: 3 to 5 days.
Acute interdigital necrobacillosis: 3 days.
Acute post-partum metritis within 10 days after calving: 5 consecutive days.
Maximum recommended dose at one injection site: 6 ml.
Special warnings
Do not mix with other veterinary medicinal products.
The product should be reserved for the treatment of clinical conditions which have responded poorly, or are expected to respond poorly to first line treatment.
National and regional antimicrobial policies should be taken into account when the product is used.
Whenever possible, the product should only be used based on susceptibility testing.
The product is intended for treatment of individual animals. Do not use for disease prevention or as a part of herd health programmes.
Do not use as prophylaxis in case of retained placenta.
Do not handle this product if you know you are sensitized.
Handle this product with great care to avoid exposure. Wash hands after use.
If you develop symptoms following exposure, seek medical advice or urgent medical attention.
Withdrawal periods
Meat and offal: 8 days.
Milk: zero hours
Shelf life
24 months
Storage
Store in a cool and dry place at (15 – 25)℃, protect from light.
Keep out of the reach and sight of children.